Thermodynamics: The Science of Heat, Work, and Energy
Thermodynamics is a fundamental branch of physics that deals with the laws governing heat, work, and energy in a system. This post will explore the core principles of thermodynamics and their application in various scientific and engineering fields.
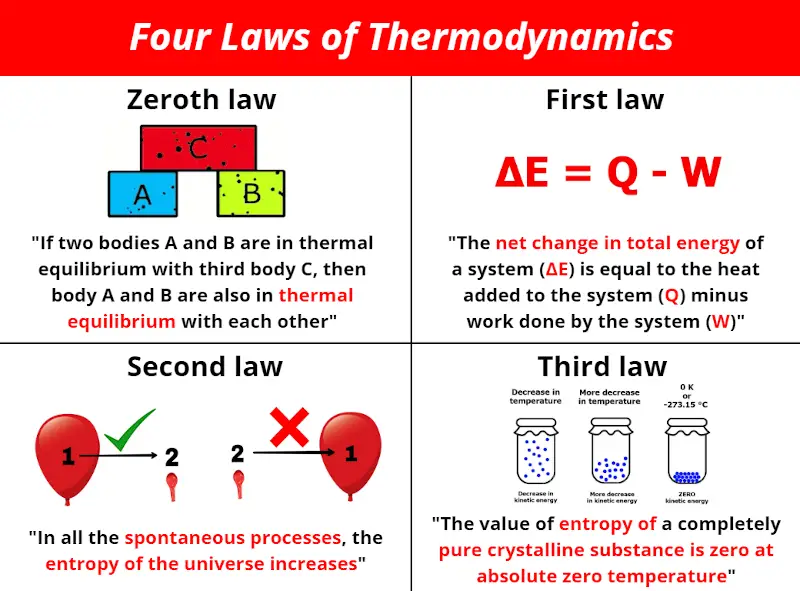
Core Principles of Thermodynamics
- First Law (Conservation of Energy): Energy cannot be created or destroyed, only transformed from one form to another. This law is often expressed as the change in internal energy of a system being equal to the heat added to the system minus the work done by the system.
- Second Law: Entropy, a measure of disorder or randomness in a system, always increases in an isolated system. This law implies that energy transformations are not completely efficient and some energy is always lost as heat.
- Third Law: As the temperature of a system approaches absolute zero, the entropy of the system approaches a constant minimum.
Applications of Thermodynamics
- Engines and Refrigeration: Thermodynamics is crucial in the design and operation of engines and refrigerators, which work on the principles of heat transfer and energy conversion.
- Chemical Reactions: In chemistry, thermodynamics helps predict the spontaneity of reactions and the equilibrium state of reactions.
- Environmental Science: It plays a role in understanding the Earth’s energy balance and climate systems.
Everyday Examples
- Heating and Cooling Systems: The heating of homes and the cooling of food in refrigerators are practical applications of thermodynamic principles.
- Power Generation: The generation of electrical power in thermal power plants involves converting heat energy into mechanical work and then into electricity.
Thermodynamics, with its laws and concepts, is not just a theoretical framework; it underpins much of the technology and natural phenomena we encounter daily. From powering engines to understanding the behavior of gases and fluids, thermodynamics is integral to the field of physics and engineering.